Solutions for Medical Device Manufacturers
Our team of experts serves medical device manufacturers by providing high-quality services for the regulatory lifecycle of your products.
Solutions tailored to your needs
Our medical device, in vitro diagnostic, and CRO experts work together to create the most efficient solutions for you. We are a team of multidisciplinary professionals with years of industry experience. Our flexible resources are ready to quickly step in to deliver prime regulatory and quality service. We always work from your unique perspective. Whether a one-time assignment or a long-term collaboration, we support you in every phase of your regulatory journey to reach your business goals.
Experience seamlessly applied to the entire regulatory life cycle of your medical device.
IVDR or MDR transition in your hands?
We provide several tools to tackle transition projects efficiently. Each transition project is different, but the process is the same:
- Identification of the gap
- Filling the gap (incl. evaluating and creating more clinical evidence when needed)
- EU registration
Digital Health
We have a dedicated DigiHealth Unit with multi-professional competence to support your journey towards sales approvals with our medical devices.
Are you expanding to new markets?
All markets have their requirements for medical devices. However, the EU CE mark or US FDA clearance/approval is sometimes sufficient evidence for market entry and sometimes an excellent start for the registration process. Just remember to keep QMS compliant.
Capacity issues?
Do you have medical devices that need to be registered to new markets, and their technical documentations need to be updated?
We can provide a professional team to work with your team as a capacity extension unit.
Medical device & IVD manufacturers
Compliance with the regulatory life cycle of your product
We provide you support starting from the first idea of the product until the end of the product life cycle.
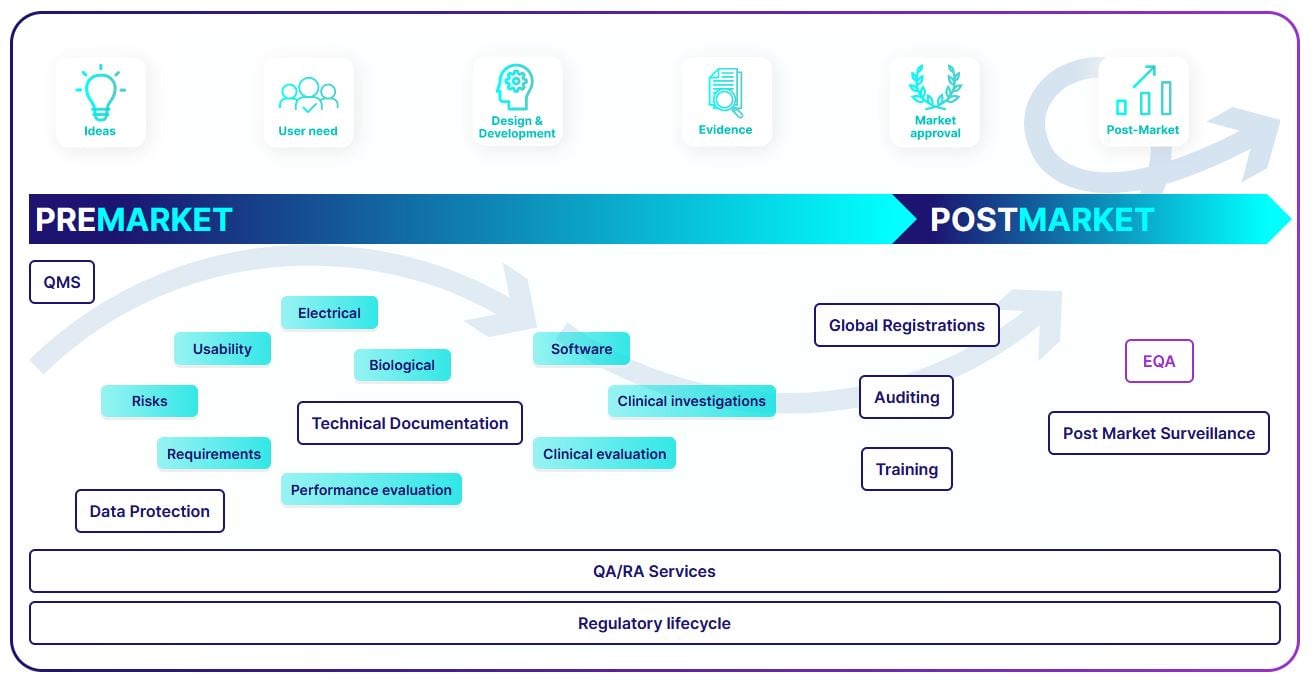
Premarket
Postmarket
Our references

Do you have an idea for a new product?
Time to contact us!
Getting a product idea is great! In a heavily regulated environment, you will thank yourself for contacting quality and regulation experts as early as possible to get the process straight and visible right away.
Keys to Success in Health Technology
Healthcare technology life cycle management is necessary to maintain patient information security, stay compliant, and improve patient care. We excel in medical device life cycle services and quality management. Discover our service portfolio below and find help for your needs today.
Market access
The services provided by Labquality will streamline market access for medical devices.
Quality Management Systems
Labquality’s experts provide assistance in building and managing quality management systems.
Device requirements
The solid know-how and experience of Labquality’s experts bring clarity to device requirements and regulations.
Training
Labquality offers a wide range of training sessions for medical device and health tech professionals.
Clinical Investigations
Clinical investigations are demanding processes, but they are also the most reliable way to produce high-quality information about the operation of a medical device. Labquality provides services and expertise in planning, preparing, and conducting clinical investigations.
IVDR compliance for in-house developed tests
The new In Vitro Diagnostic Medical Device Regulation (IVDR) will be applied in phases starting on 26 May 2022. The IVDR and the national law of each EU Member State also concern in vitro diagnostics (IVDs) developed in-house by clinical laboratories.

Contact us for more information
Leave a contact request
Explore our services
The solid know-how and experience of Labquality’s experts bring clarity to device requirements and regulations.

Technical documentation
We can support you in getting your Quality Management System and Technical Documentation compliant with the regulations.

Regulatory Life cycle
We help medical device companies in meeting the new, comprehensive requirements and compliance expectations during the entire lifecycle of their products.

Regulatory Essentials in Health Tech
This training series provides an efficient introduction to the regulatory essentials to turn regulatory compliance into business advantage.

Clinical Investigation
Labquality provides services and expertise in the planning, preparation, and conduct of clinical investigations.